Filipa SIOPA
Sorbonne Université, France
Imine Furfurals for Ruthenium-Catalyzed Direct C3-H Arylations and Alkenylations
Implementation of catalytic C-H transformations on renewable feedstock is becoming an attractive strategy for green transformations. In particular, lignocellulosic biomass valorization has received significant attention as an alternative to the limited petroleum resources. Furfural and 5-(hydroxymethyl)furfural (HMF) are among the most important unsaturated large-volume chemicals that can be directly prepared from biomass. Although some transition metal catalyzed direct C-H functionalizations of furfural have been reported, only very few address the selective C3-H functionalization.
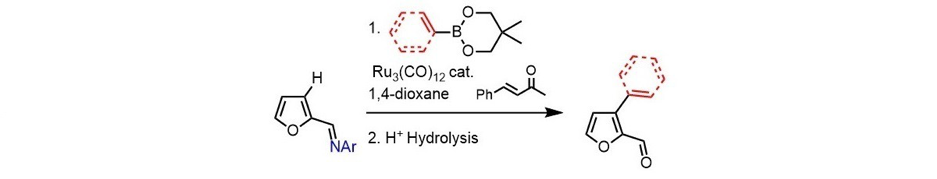
Herein, we describe the Ru(0)-catalyzed C3-H arylation and alkenylation of furfural imines with boronate derivatives, followed by mild acid hydrolysis, to access the arylated- or cinnamylated-furfural derivatives (Scheme 1).
Acknowledgements: The authors would like to acknowledge Horizon 2020 ERANet-LAC project CelluloseSynThech for financial support, as well as CNRS, Sorbonne Université and Labex Michem (Investissements d’Avenir programme, ref. ANR-11-IDEX-0004-02). Support through CMST COST Action, CA15106 (CHAOS) and the Fundação para a Ciência e Tecnologia (SFRH/BPD/88666/2012) is also gratefully acknowledged.
1. F. Roudesly, J. Oble, G. Poli, J. Mol. Cat. A. 2017, 426, 275.
2. J. S. Luterbacher, D. M. Alonso, J. A. Dumesic, Green Chem. 2014, 16, 4816; T. A. Bender, J. A. Dabrowski, M. R. Gagne, Nature Reviews Chemistry 2018, doi:10.1038/s41570-018-0005-y.
3. a) R. Mariscal, P.Maireles-Torres, M. Ojeda, I. Sádabaa, M. López Granados, Energy Environ. Sci. 2016, 9, 1144. b) A. A. Rosatella, S. P. Simeonov, R. F. M. Frade, C. A. M. Afonso, Green Chem. 2011, 13, 754.
4. a) A. Jakab, Z. Dalicsek, T. Soos, Eur. J. Org. Chem. 2015, 56. b) X. Jie, Y. Shang, P. Hu, W. Su, Angew. Chem. Int. Ed. 2013, 52, 3630. c) Y. Shang, X. Jie, J. Zhou, P. Hu, S. Huang, W. Su, Angew. Chem. Int. Ed. 2013, 52, 129. d) X. Jie, Y. Shang, P. Hu, W. Su, Angew. Chem. Int. Ed. 2013, 52, 3630.
5. C. Pezzetta, L. F. Veiros, J. Oble, G. Poli, G. Chem. Eur. J. 2017, 23, 8385.
6. a) F. Hu, M. Szostak, Org. Lett. 2016, 18, 4186. b) F. Siopa, V. A. R. Cladera, C. A. M. Afonso, J. Oble, G. Poli. Eur J Org Chem 2018, Accepted. doi: 10.1002/ejoc.201800767
