Neringa Tamosiunaite
University of Leicester, United Kingdom
Electronic and Steric Effects on C-H Activation
In recent years metal catalysed C-H has become an important tool in synthesis of pharmaceuticals, biological molecules and natural products.1 To expand the scope further it is crucial to understand the mechanism(s) behind these transformations. Two similar mechanisms have been proposed for carboxylate assisted C-H activation: Ambiphilic Metal Ligand Activation (AMLA)2 and Concerted Metallation-Deprotonation (CMD).3 AMLA favours electron donating groups (EDG) in cyclometallation reactions,4 whilst CMD in some cases favours electron withdrawing groups (EWG) in direct arylation.3 The reasons for this difference are not clear but could be due to use of different substrates and/or metals.
This work aims to study electronic effects on C-H activation via competition between two substituted phenylpyridines at Ir and Rh (Figure 1). The ratio of 1 and 2 at early reaction times reflects relative rate of formation of two products. If the reactions are reversible and ratio of 1:2 changes that shows thermodynamic selectivity.

Figure 1: Study of electronic effects on C-H activation
The results show that EDGs are kinetically preferred for both metals which is consistent with results observed for AMLA. For Ir no change in the product ratios were observed. Whereas for Rh the ratios change over time to favour EWGs indicating that C-H activation with Rh is reversible.
Steric effects will be compared to Jones earlier work with phenylpyridines substituted at 3 position that after cyclometallation with metal (Ir, Rh) can give two regiosiomers: ortho and para (Figure 2).5 The results show that sterically less crowded R = OMe with Ir and Rh produces both regioisomers in similar ratios whilst for bigger R = CF3 the para selectivity is much more pronounced and observed in ≥1:10 ortho:para ratio.
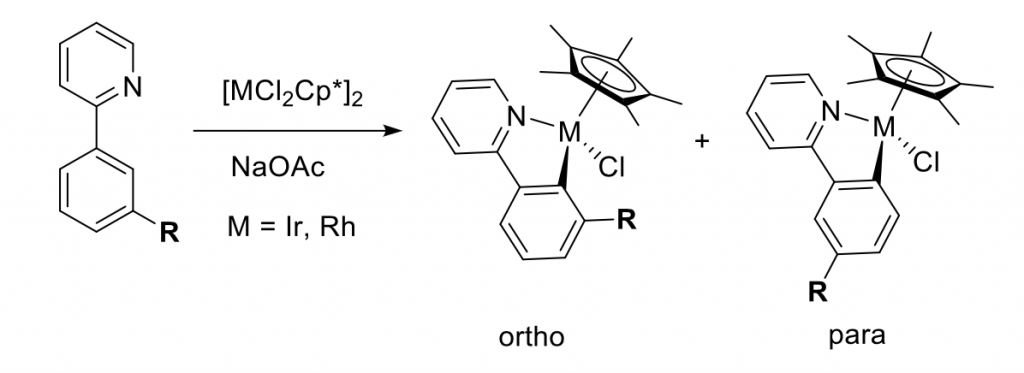
Figure 2: Study of steric effects on C-H activation
- J. Yamaguchi, A. D. Yamaguchi and K. Itami, Angew. Chem. Int. Ed., 2012, 51, 8960-9009.
- Y. Boutadla, O. Al-Duaij, D. L. Davies, G. A. Griffith and K. Singh, Organometallics, 2009, 28, 433-440.
- S. I. Gorelsky, D. Lapointe and K. Fagnou, J. Am. Chem. Soc., 2008, 130, 10848-10849.
- A. D. Ryabov, I. K. Sakodinskaya and A. K. Yatsimirsky, J. Chem. Soc., Dalton Trans., 1985, 2629-2638
- L. Li, W. W. Brennessel and W. D. Jones, Organometallics, 2009, 28, 3492-3500.
