Hydrosilylation of carbonyl compounds using well-defined PNP-Mn(I) hydride complexes
Latest Posts
-
6th CHAOS meeting in Cyprus from 3.4. to 5.4.2019-ReminderDec 19
Dear CHAOS Members, So far the number of registrations for Cyprus meeting is low as most of the members have…
-
Tarragona meeting scientific program is onlineSep 10
Dear CHAOS Members, Tarragona meeting scientific program is online and can be viewed here, http://www.events.cost-chaos.org/program/#not-set:all We could still fit couple…
-
Early Bird Registration is now available for CHAOS membersJun 10
Registrations open for CHAOS meeting in Tarragona Spain 19.9.-21.9 CHAOS meeting in GP3 at the ICIQ in Tarragona Spain Meeting…
Stefan Weber
Institute of Applied Synthetic Chemistry, TUWien
The reduction of carbonyl compounds is among the most important transformation in the field of organic chemistry. This reaction can be performed with hydrogen and a transition metal catalysts, based on precious metals as well as base metals1. Especially well-defined manganese (I) compounds are of great interest for these transformations2, since manganese is as an abundant resource. Although hydrogen is the cheapest of all reduction reagents and therefore frequently used, it has several drawbacks. Apart from being a flammable gas, (high)-pressure apertures are needed in order to perform reactions.
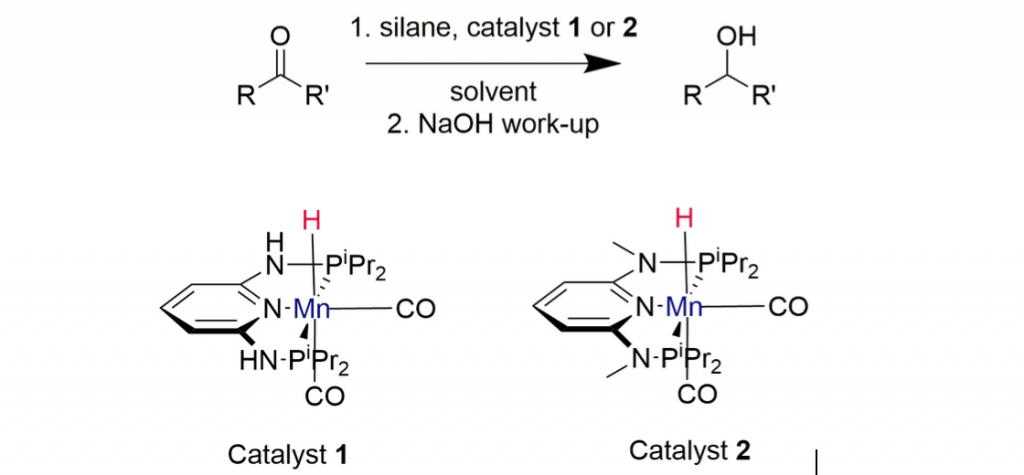
Figure 1. Reaction scheme of hydrosilylation
The role of silanes were investigated as possible hydrogen donors for the reduction of ketones and aldehydes. This procedure allows catalytic reduction in various solvent under mild reaction parameters, using well-defined PNP-Mn(I) hydride complexes. Apart from Phenylsilane as hydrogendonor, the inexpensive Polymethylhydrosilane (PMHS) can be used with catalyst loading below 2 mol% to achieve reduction of ketones and aldehydes.
References
(1) Gorgas, N.; Sto, B.; Veiros, L. F.; Pittenauer, E.; Allmaier, G.; Kirchner, K. Organometallics 2014, 33, 6905–6914.
(2) Garbe, M.; Junge, K.; Beller, M. European J. Org. Chem. 2017, 2017 (30), 4344–4362.
