Copyright ©2017 all rights reserved
Designed by Plethora Themes
Designing New Building Blocks: Incorporation of Electron Rich Thiophene into the Fully Planar Indolo[3,2,1-jk]carbazole Scaffold
Institute of Applied Synthetic Chemistry, TUWien, Austria
Triarylamines (TAAs) represent essential building blocks as electron donors in the field of organic electronics (e.g. organic light emitting diodes) [1], [2]. Electronic properties such as triplet energy ET, HOMO and LUMO levels as well as the donor strength of the TAAs can be tuned by planarization of the molecular scaffold and the incorporation of heteroatoms. As previously shown by our group, increased planarization leads to decreased donor strength and the fully planar indolo[3,2,1-jk]carbazole (ICz) (Figure 1) even exhibits weak acceptor characteristics [3], [4].
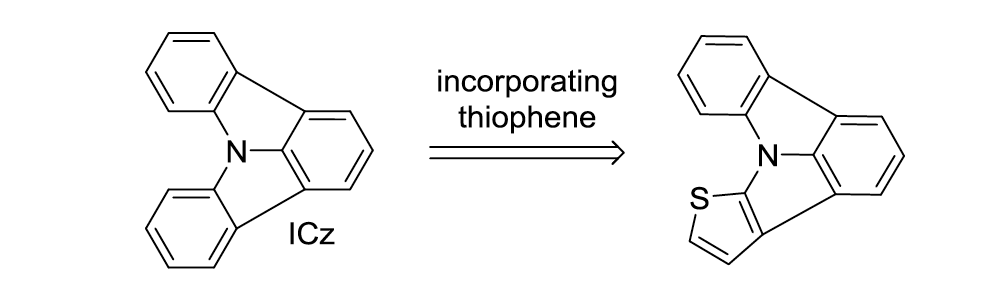
Figure 1: Schematic representation of the incorporation of thiophene into the ICz scaffold
The aim of our current research is to increase the donor strength of the fully planar ICz by introducing electron rich thiophene units into the ICz scaffold. A synthetic approach applying Ullmann condensation as well as CH activation will be presented in this contribution. Furthermore, the photophysical and electrochemical characterizations as well as theoretical calculations of the newly developed target molecules will be discussed.
____
[1] Y. Shirota and H. Kageyama, “Charge Carrier Transporting Molecular Materials and Their Applications in Devices,” Chem. Rev., vol. 107, no. 4, pp. 953–1010, Apr. 2007.
[2] Y. Tao, C. Yang, and J. Qin, “Organic host materials for phosphorescent organic light-emitting diodes,” Chemical Society Reviews, vol. 40, no. 5, p. 2943, 2011.
[3] P. Kautny et al., “Oxadiazole based bipolar host materials employing planarized triarylamine donors for RGB PHOLEDs with low efficiency roll-off,” Journal of Materials Chemistry C, vol. 2, no. 11, p. 2069, 2014.
[4] P. Kautny et al., “Indolo[3,2,1-jk]carbazole based planarized CBP derivatives as host materials for PhOLEDs with low efficiency roll-off,” Organic Electronics, vol. 34, pp. 237–245, Jul. 2016.