Copyright ©2017 all rights reserved
Designed by Plethora Themes
Ru-catalyzed carbonylative Murai reaction of N-containing aromatic systems
Università dell’Insubria, Italy
Nowadays, the direct and selective functionalization of non-acidic C-H bonds has emerged as a novel step-economical and eco-compatible strategy. It allows the use of simple and cheap starting materials for the construction of complex organic molecules by adding the C-H bonds to the most common directing and functionalizable groups, such as carbonyls, halides, alcohols, etc.
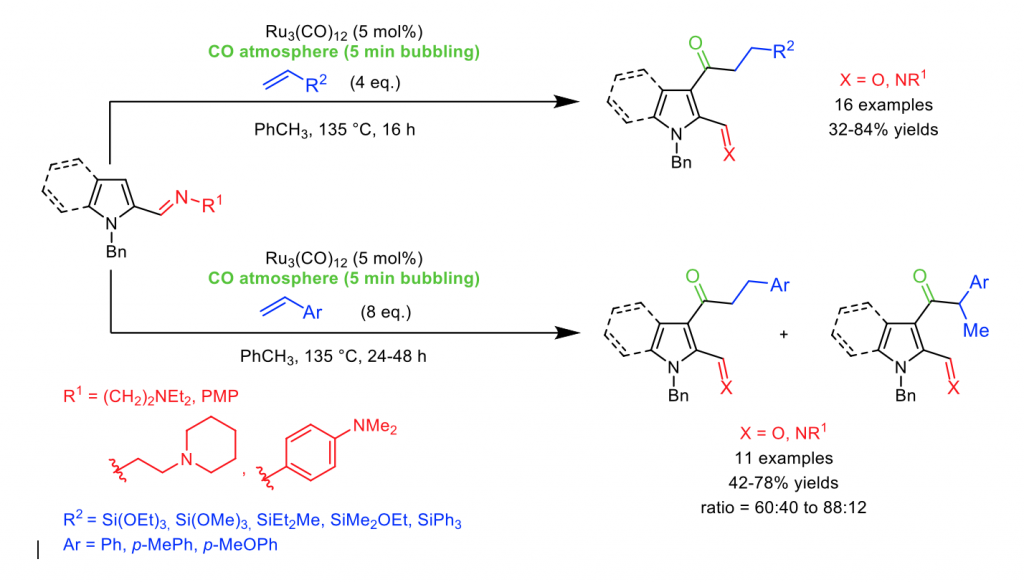
Furthermore, the direct functionalization of nitrogen containing heterocycles, moieties commonly present in many natural products, drugs and enzymes, remains an important topic.[1] In this context, we focused on a direct C3-H functionalization of nitrogen containing rings – such pyrrole or indole –, via a carbonylative version of the well-known Murai reaction.[2]
Basing on previous line research of our group,[3] we have recently developed a directed Ru(0)-catalyzed C3-acylation of N-heterocycle (pyrrole/indole) imines, which can be obtained by treatment with vinylsilanes or styrenes under carbon monoxide atmosphere. Various 3-carbonylated pyrrole- or indole-imines were obtained with moderate to good yields, as well as the corresponding aldehydes after hydrolysis, In the case of styrenes, a mixture of the linear (major) and branched (minor) isomers was obtained. In this communication, discovery, optimization and substrate scope of this new C-H activation based transformation will be disclosed.
[1] (a) KulKarni, S. K.; Singh, V. P. Curr Top Med Chem., 2007, 7, 251-263. (b) Joule, A. Indole and its Derivatives in Science of Synthesis: Houben-Weyl Methods of Molecular Transformations, Vol. 10 (Ed.: Thomas, E. J.), Thieme, Stuttgart, 2000; Chapter 10.13.
[2] Murai, S.; Kakiuchi, F.; Sekine, S.; Tanaka, Y.; Kamatani, A.; Sonoda, M.; Chatani, N. Nature 1993, 366, 529-531
[3] Pezzetta, C.; Veiros, L. F.; Oble, J.; Poli, G. Chem. Eur J., 2017, 23, 8385-8389.