
Efficient Henry addition by a (Sb)G graphene catalyst
Vaseli Parvulescu
University of Bucharest, Bucharest, Romania
Graphene, an allotrope of carbon, has been considered as an excellent suitable catalytic support due to its high carrier mobility, high electrical and thermal conductivity, high surface area, unique two-dimensional (2D) honeycomb lattice, high electron mobility, strong metal-support interactions and easy surface functionalization [1]. In addition, doping graphene-nanosheets with heteroatoms became an attractive method of tuning its electronic and catalytic properties [2]. In this context, we recently demonstrated that coupling reactions can be efficiently catalyzed by MNPs strongly grafted on few- and multilayer defective graphenes prepared by pyrolysis of natural polysaccharides containing metal salts [3]. In order to prove the versatility of the graphenes for coupling reactions in this study we extended the investigation of these materials using Sb as doped heteroelement of graphene and Henry addition as a model reaction (Scheme 1).
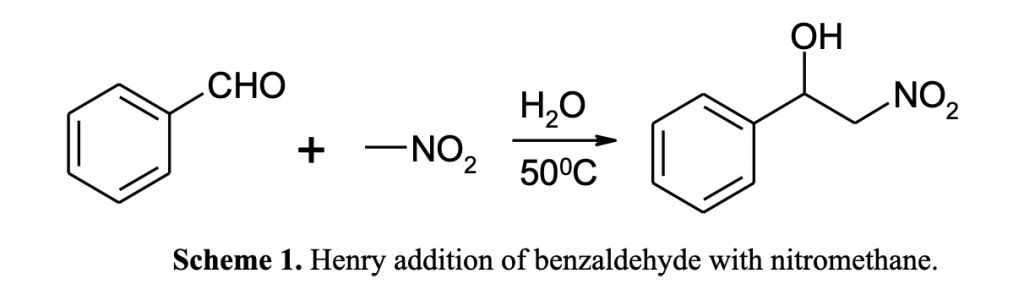
The catalysts were exhaustively characterized using XRD, Raman, XPS, and DRIFT spectroscopy. Activity tests were carried out in a glass reactor containing 3 mL of solvent (dried isopropyl alcohol (IPA) or deionized H2O) with benzaldehyde (1-0.5 mmoles), nitromethane (10mmoles) and the catalyst. The resulting mixture was stirred for 2-24 h at room temperature or 50ºC. The recovered products from the liquid phase were analyzed and identified by GC-MS and 1H and 13C NMR spectroscopy.
These experiments confirmed (Sb)G as a highly efficient catalyst for the coupling of benzaldehyde with nitromethane to nitoaldol. In water, it allowed a 42% selectivity to nitroladol for a total conversion of benzaldehyde. Optimal catalytic features and reaction conditions as well as the catalytic performances – catalytic properties correlation with the catalytic results will be discussed in detail.
Acknowledgements
The authors kindly acknowledge UEFISCDI (PN-III-P4-ID-PCE-2016-0146, nr. 121/2017) for the financial support.
References
1. G. Bottari, M. Á. Herranz, L. Wibmer, M. Volland L. Rodríguez-Pérez, D. M. Guldi, A. Hirsch, N. Martín, F. D’Souza, T. Torres, Chem. Soc. Rev., 2017, 46, 4464-4500.
2. A. Bostwick, F. Speck, T. Seyller, K. Horn, M. Polini, R. Asgari, Science, 2010, 328, 999-1002.
3. A. Primo, I. Esteve, J. F. Blandez, A. Dhakshinamoorthy, M. Alvaro, N. Candu, S. M. Coman, V. I. Parvulescu, H. Garcia, Nat. Commun. 2015, 6, 8561.
